Bacteria’s Explosive Secret Revealed
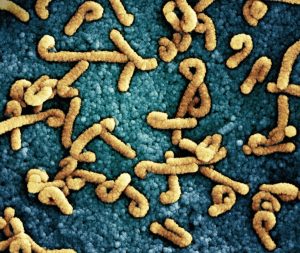
Researchers have uncovered a startling survival mechanism that allows bacteria to share life-saving genetic information—including antibiotic resistance—by literally exploding. In a study published this month, scientists identified a specific group of three genes, known as LypABC, that acts as a command center for this process. When activated, these genes force the bacterial cell to burst, releasing tiny, virus-like particles called Gene Transfer Agents (GTAs) that act as couriers to spread DNA to neighboring microbes.
For years, microbiologists have puzzled over how bacteria rapidly develop resistance to modern medicine. While some bacteria share DNA through direct contact, this new discovery explains a far more aggressive method. These GTAs are essentially ancient viral invaders that bacteria have tamed and repurposed over eons. Instead of causing an infection, the bacteria now use these particles as a biological mail service to trade genetic traits, including the ability to withstand life-saving antibiotics.
The Mechanics of Microbial Warfare
The LypABC gene cluster serves as the “on switch” for this cellular suicide mission. When conditions demand it, the bacteria trigger their own destruction to ensure the survival of the colony. By exploding, the cell releases a flood of GTAs packed with genetic instructions. Nearby bacteria then absorb these particles, incorporating the new DNA into their own genetic makeup. This allows a community of bacteria to gain immunity to drugs collectively, even if only a few cells were initially exposed to the threat.
This discovery provides critical insight into why antibiotic-resistant “superbugs” are so difficult to eradicate. If bacteria can communicate and share defensive strategies through these explosive bursts, traditional medicine faces an uphill battle. The study highlights that these microscopic organisms are far more sophisticated and collaborative than previously thought, turning what was once a viral threat into a tool for their own evolutionary advantage.
Understanding this mechanism could change how we approach infectious disease. By identifying the LypABC cluster, scientists are now looking for ways to “jam the signal” or block the release of these genetic couriers. If researchers can prevent the bacteria from sharing their resistance, existing antibiotics might once again become effective against stubborn infections. However, the complexity of these biological systems serves as a reminder of the intricate design found in even the smallest forms of life, challenging us to continue seeking wisdom and ingenuity in the face of evolving health challenges.